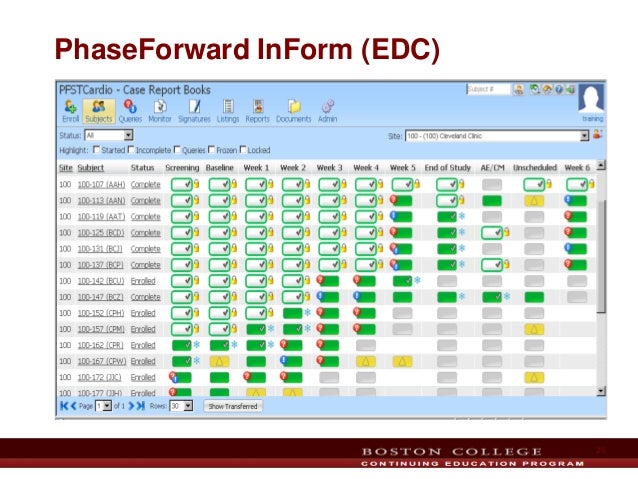
However that change was well received and soon became the way of life for the site users. There were jitters when the traffic lights were replaced with colored boxes starting v5.0. It had the most popular traffic lights features till v4.6. It's not that InForm UI itself didn't change. To name a few of them - VieDoc, Caster EDC, Medrio have taken the UI to the next level and have made backend programming a lot less technical. The backdrop is of other interesting EDCs that have crowded the scene earlier dominated by InForm and Rave. 
It will tender it just that dry and bare - Phone. If this is true, then it amounts to InForm becoming OutForm in the new offering. (Please share your impressions in the comments section for good)ġ) Clinical One does not deploy to InForm I have gathered mixed responses in last couple of months and I share them here as they are. So I return to the original speculation of whether InForm is being retired? (the backend does add power to the UI, but site users just see the UI) And that's what matters most to the site users. I started with InForm 4.0 way back in year 2005 and till date, I come across site users (and sponsor users, of course!) who simply take InForm as the de facto eCRF solution, thanks mainly to the great UI. However what about the end user community of InForm loyals - mainly the site users? And it's a welcome change for the CRF designers and developers community. 
The idea of Clinical One is really innovative and self-service platform based. I spoke to former Phase Forward and Oracle colleagues and the excitement is not unfounded. 
Like many of you, I have been attending to online fora where Clinical One is being discussed with lot of excitement. The jump start can be completed within ~8 weeks.ĭoes your organization face similar study-building challenges? Do you have any other Oracle InForm initiatives that are coming up for which you may need to rely on an experienced team of clinical data management and IT subject matter experts? If yes, please reach out to me for more information.That's a question doing rounds in many minds including mine and this is my attempt to speculate on it either ways. With our Oracle InForm Study-in-a-Box jump start, our clinical data management and EDC experts can efficiently build your InForm studies. Multiple studies built in compliance with the company’s SOPsĪllowed sponsor to be nimble and supplement its team without hiring additional people Resources from our InForm build team assisted with study builds, including library development and template creation Provided dedicated resources to assist company with process re-engineering The company needed to supplement its study-building capabilities with outside support. The first project I want to share with you is one that we did for a manufacturer of pharmaceuticals that uses InForm as its electronic data capture and clinical data management solution to collect data on each patient participating in a clinical trial. Providing resources to build and support clinical studies in InForm If you walk away knowing a little more about us, I’ll consider it a win. Or, perhaps you don’t need assistance in these particular areas but weren’t aware that we have experience with InForm. Maybe you’ve been struggling with similar challenges and weren’t sure who to turn to for help. While each company has its own priorities and initiatives, I believe it’s worth sharing a recent InForm project that we’ve completed, as well as two offers that might interest you. Learn how AI/ML can be used by pharmaceutical and medical device companies to improve the clinical data review and cleansing process. How Artificial Intelligence Can Enhance the Clinical Data Review and Cleaning Process
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
